Research
Learning across the lifespan: from behavior to circuits
mission
Humans and animals continuously acquire, adapt, and maintain a rich set of sensory-guided behaviors, learning to retain and reuse them across the lifespan. In health, we balance learning new behaviors with preserving or adjusting existing ones. In aging and disease, we may struggle to recall familiar knowledge, raising the question of whether memories are lost or simply dormant.
.avif)
The Kuchibhotla Lab explores the neural mechanisms that enable lifelong learning and flexibility, from single-task learning and habit formation to naturalistic forms of continual, multi-task learning. Our studies have uncovered phenomena that prompt the field to revisit canonical principles about how quickly learning occurs, when habits emerge, and why memory fails. Starting in animal models and the auditory system, we deploy novel behaviors, longitudinal neural recordings, and causal perturbations to generate rich data sets across cell types, circuits, and regions. We use these insights to build models of flexible learning and to reverse engineer disruptions seen in aging and neurodegeneration.
Approach
Behavior

principles

circuits
We begin with behavior. We build scalable tasks—from single-task learning and habit formation to continual, multi-task training in naturalistic home cages—so animals can learn over weeks to months. We are deliberately pluralistic: head-fixed when we need millisecond control and stable imaging; freely moving when context, navigation, or social interaction matter. We pair tightly controlled assays with naturalistic paradigms, always choosing the simplest preparation that answers the question. This design-first philosophy lets us test how knowledge is built, preserved, reused, or disrupted and sets the stage for clear conceptual models and neural interrogation.
From these observations, we articulate simple, testable principles for flexible learning. We explore literature across disciplines to gain insight into questions about learning versus performance (cognitive science), compositionality and continual learning (machine learning), and habit formation (learning theory). We treat neurodegeneration-related memory deficits as a conjunction of memory loss and retrieval deficits, allowing us to explore both possibilities. These conceptual models guide the next experiment (and vice versa in iterative loops), and we formalize them with theorist partners when math sharpens or falsifies the idea.
We then probe mechanisms in neural circuits. Using longitudinal optical approaches (two-photon imaging, miniscopes, fiber photometry) and targeted perturbations (opto- and chemo-genetics) during behavior, we follow identified cell types and distributed networks—the central auditory system, posterior parietal cortex, medial prefrontal cortex, and striatum—as animals learn. By isolating circuit-level mechanisms and coding principles that support flexible learning, we can ask what fails in aging and neurodegeneration and then design interventions to reverse engineer healthy function.
research directions
Neural circuit mechanisms of learning versus performance

Animals often ‘know’ a rule before they can show it. Learning is a relatively durable change in capability, while performance is the moment-to-moment expression of that capability and is shaped by exploration, strategy, motivation, fatigue, and context. This distinction is widely appreciated in psychology and related fields, but in systems neuroscience performance under reinforcement is still often treated as the proxy for learning.
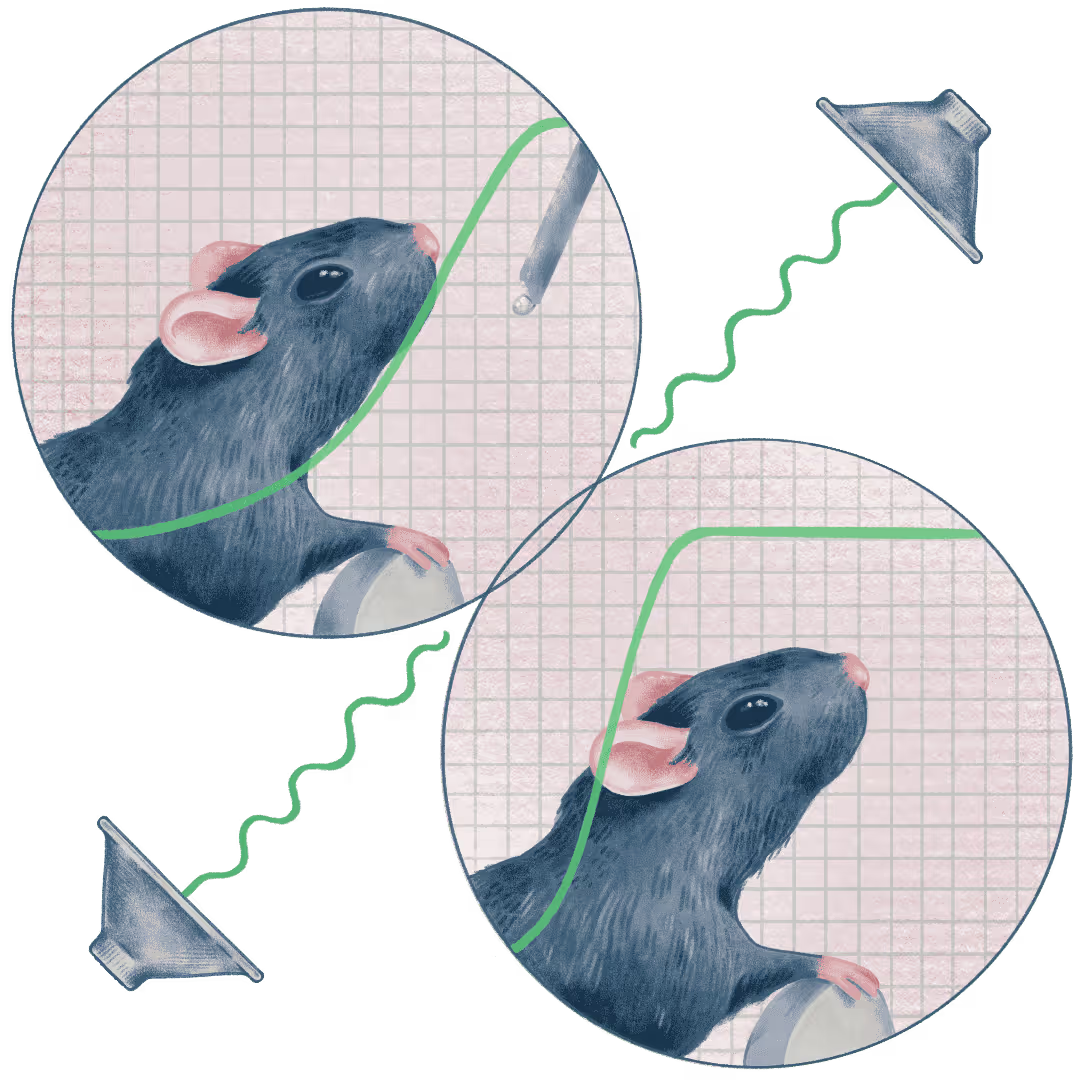
Our lab uses this dissociation as an entry point to better understanding the neural basis of learning and performance. We design behavioral paradigms and analyses that separate learning from performance, then apply modern circuit tools to ask what changes in the brain while behavior still looks “unlearned,” and what mechanisms convert latent knowledge into reliable, stable performance. In a sound-guided discrimination, we have found that rodents extract task contingencies much earlier than previously assumed, followed by a long phase of structured exploration that gradually refines performance over hundreds to thousands of trials.
We combine longitudinal two-photon imaging, targeted optogenetic perturbations, and sophisticated analyses (including tensor decomposition) to identify circuit computations supporting rapid learning versus stable performance. Using these approaches, we showed that auditory cortex can drive rapid learning and consistent performance through non-canonical higher-order mechanisms. We are now defining how cholinergic and noradrenergic pathways reshape learning and performance across timescales, down to microcircuit interactions among neuromodulatory inputs, excitatory/inhibitory neurons, and astrocytes.
Finally, we find that the involvement of sensory cortex evolves with practice: early on, auditory cortex provides a teaching signal; with extended training, control appears to shift toward subcortical auditory (inferior colliculus, auditory thalamus) and non-auditory (striatum) circuits. By combining two-photon mesoscopic imaging, causal perturbations, and modeling, we aim to define the mechanisms of this cortico–subcortical tutoring process across much of the central auditory network.
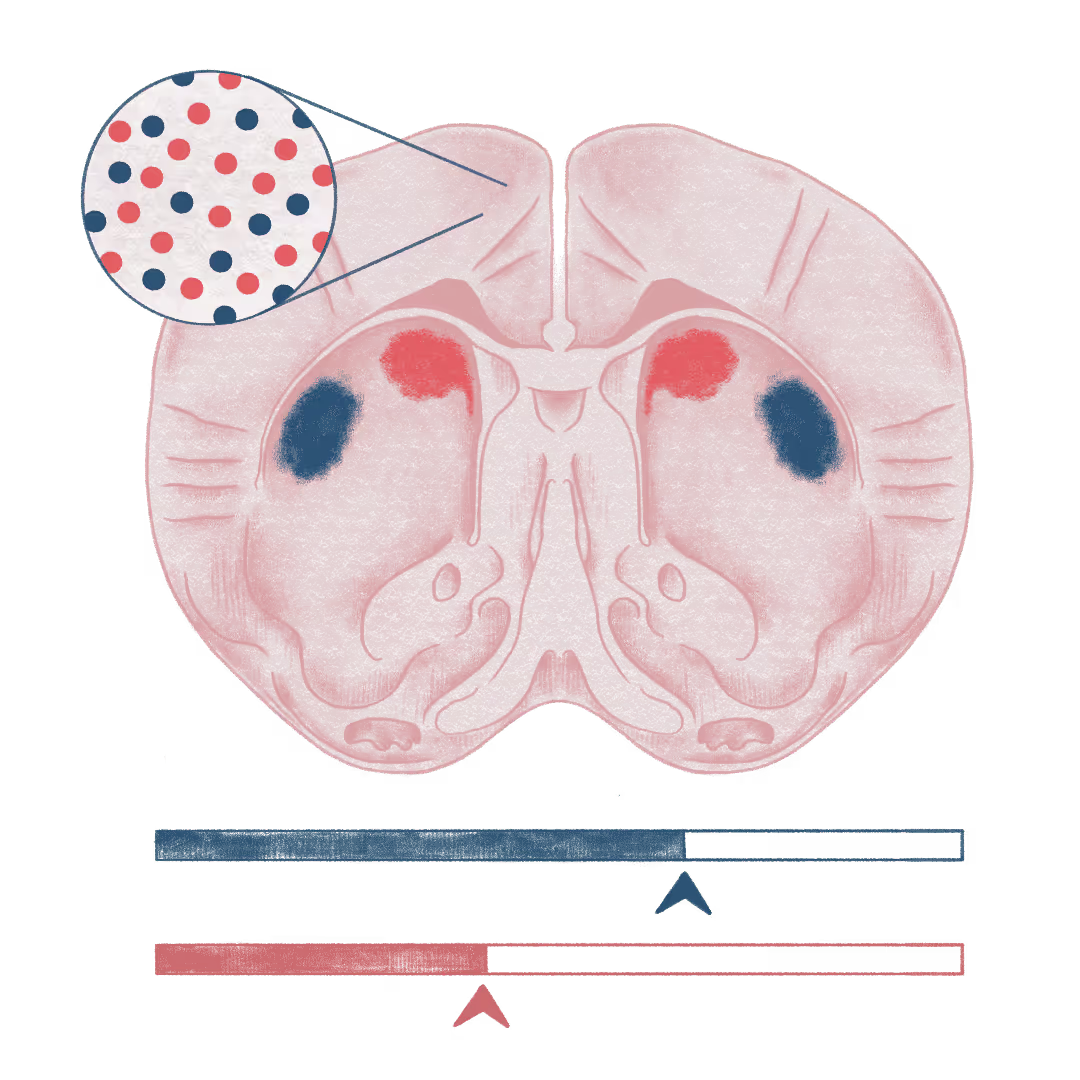
Corticostriatal control of abrupt transitions from goal-directed to habitual behavior

Habit formation is hard to study in real-time because its onset is usually invisible: by the time you “notice” a habit, it is already established. Goal-directed control is flexible and outcome-based, whereas habitual control is automatized, stimulus-driven, and can persist even when outcomes change. The prevailing view is that the shift from one to the other is gradual. Our recent work using a novel behavioral approach suggests a different picture: habit emerges as a switch-like change in control, with abrupt behavioral signatures and neural dynamics.
Multiple circuits contribute to goal-directed and habitual behavior, including the dorsal striatum. A prevailing model assigns dorsomedial striatum (DMS) to goal-directed control and dorsolateral striatum (DLS) to habit, with a slow practice-dependent handoff. We propose a different mechanism: both controllers emerge in parallel throughout learning, and projection-specific signals from premotor and prefrontal cortex bias control toward goal-directed or habitual modes. Importantly, this is not winner-take-all. We hypothesize that behavior on any given trial reflects coordination between goal-directed and habitual controllers, with their relative weighting shifting with internal state and context (e.g., uncertainty, motivation). This framework naturally allows transitions to be sudden and even endogenously reversible.
To test this model, we take a comparative approach in mice and rats and combine projection-specific two-photon imaging, fiber photometry, targeted optogenetic perturbations, and computational behavioral modeling to identify the cortical–striatal signals that track and control transitions to habit. More broadly, we think the traditional “either-or” framing is too coarse. In everyday life, habits can initiate an action while goals still shape their endpoint: for example, you automatically reach for where the coffee usually sits, but immediately adjust when it’s not there. Capturing this mixed control is central to understanding how adaptive habits support efficient behavior and how maladaptive habits persist.
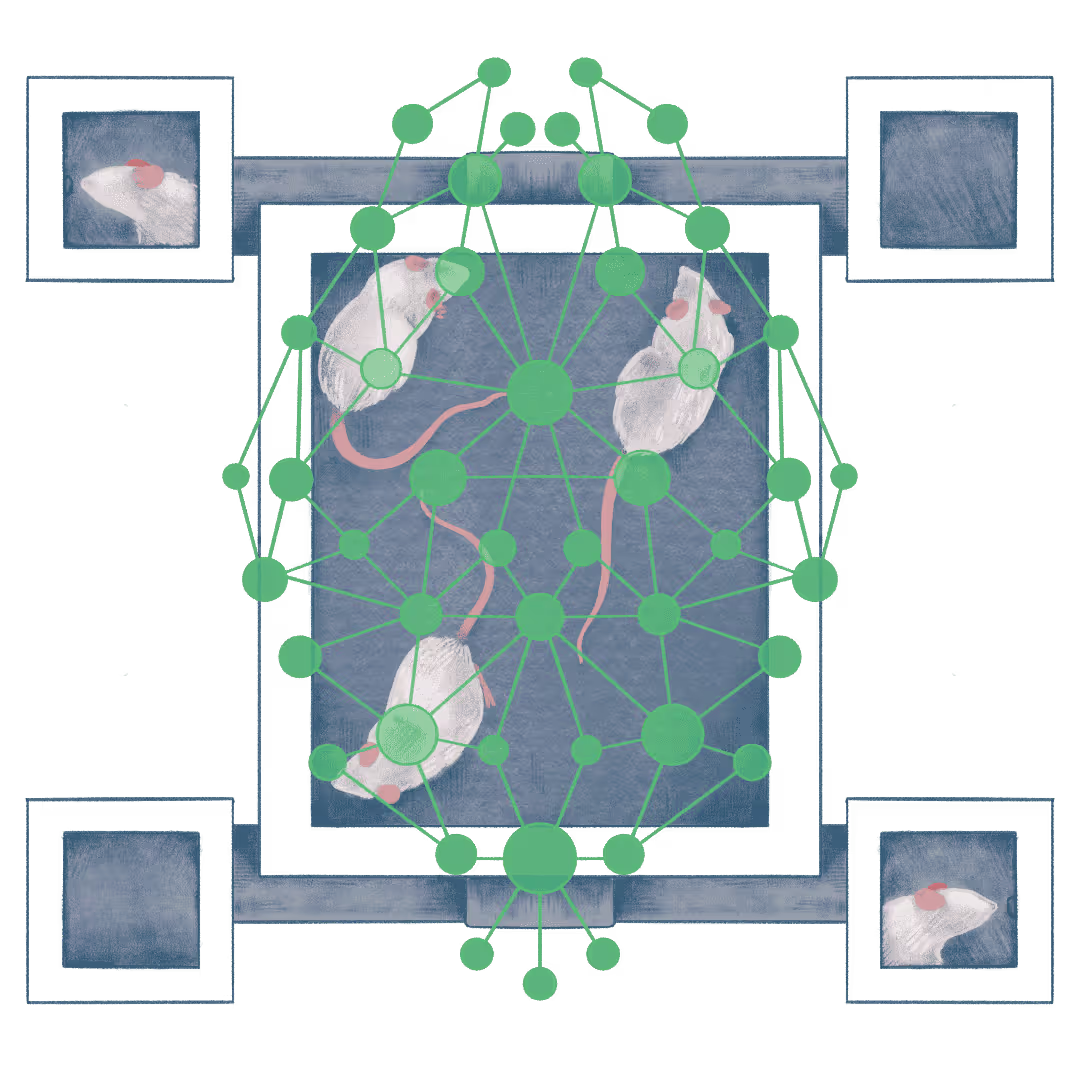
The neural basis of continual, lifelong learning

Biological brains learn throughout life by reusing old knowledge to learn new tasks by generalizing, inferring, and composing skills across contexts. This flexibility is still difficult for artificial systems, which often struggle with brittle transfer and interference when learning sequentially. We use this gap as an opportunity to ask a mechanistic question: how do neural circuits preserve what’s been learned while remaining plastic enough to acquire new behaviors?
We study continual learning with a novel, scalable “mouse playground” that enables long-term, high-throughput training, where mice learn many sensory-guided tasks over weeks to months. This platform lets us measure not just whether animals learn, but how learning transfers across task structures, when animals can infer rules, and when they can combine prior knowledge compositionally to solve new problems. We pair this behavioral approach with mini2P and benchtop-2P neural recordings and causal perturbations to track and test population-level representations across time, identifying circuit mechanisms that support inference, transfer, and compositionality.
In parallel, we collaborate on computational modeling—comparing RNN architectures that succeed or fail at continual learning, and using Bayesian/normative frameworks to formalize what efficient lifelong learning should look like—so we can align circuit mechanisms with principled algorithms.
Retrieving misplaced memories in Alzheimer’s disease

Episodes of paradoxical lucidity raise a provocative possibility in Alzheimer’s disease: some ‘lost’ memories may be stored but temporarily inaccessible. These brief returns of clarity suggest a failure of retrieval (or state) rather than complete loss of the underlying memory trace and they offer a rare window into mechanisms that can potentially restore normal cognition.
Our goal is to distinguish impairments in contextual retrieval from degradation of stored memory by identifying the circuit conditions that enable this transient cognitive recovery. In AD mouse models, we have established a new behavioral paradigm that points to cortical deficits in contextual retrieval rather than erasure of the memory itself. We are now testing how endogenous fluctuations in ascending neuromodulation and inhibition can gate access to stored memories, and whether internally driven network state shifts can unlock retrieval.
In parallel, we are building a real-world patient-tracking platform to quantify cognitive fluctuations and lucid intervals in everyday settings. Our app-based approach captures the timing, features, and potential triggers of lucid episodes through patient–caregiver reporting, cognitive testing, and wearables. This creates a scalable dataset that can be linked to mechanistic hypotheses from animal models. Together, these cross-species studies aim to reveal systems-level mechanisms that can restore access to hidden memories and to highlight therapeutic targets that could make such recovery more reliable and lasting.

Higher-order functions of the central auditory system in echolocating bats

Echolocating bats push the limits of auditory cognition—operating with extraordinary speed, precision, and robustness. By comparing bat and mouse auditory circuits, we aim to separate general principles of auditory computation from species-specialized solutions, and to identify protective mechanisms for age- and noise-related hearing loss that could be translated into more vulnerable systems.
Bats rely on sound to act in the world: they emit calls, parse echoes, distinguish self-generated from external sounds, track objects and conspecifics, and update their own actions tens of times per second. They are also unusually resilient with hearing remaining strong in noise and often across aging, making them an “upper-bound” model for what auditory systems can achieve. This creates a rare opportunity to ask not only how an auditory system can be fast and adaptive, but also what makes it stable and robust.
Together with the JHU Bat Lab (PI: Cynthia Moss), we ask three comparative questions across bats and mice in the auditory midbrain, thalamus, and cortex: (1) which computations are conserved versus species-specific, (2) how corticofugal loops rapidly retune subcortical processing, and (3) what circuit/cellular programs confer noise- and age-resilience. A long-term goal is to reverse-engineer these protective motifs into model systems (and eventually humans) where hearing and auditory learning break down.
